Describe the Modern Model of the Atom
Almost all the mass of an atom is defined by its nucleus. If we drew the atom to scale and made protons and neutrons a centimeter in diameter then the electrons and quarks would be less than the diameter of a hair and the.

Timeline Of The Development Of Atomic Theory Atomic Theory Atom Timeline Project
The solution of the wave equation brings the idea of shells sub-shells and orbitals.

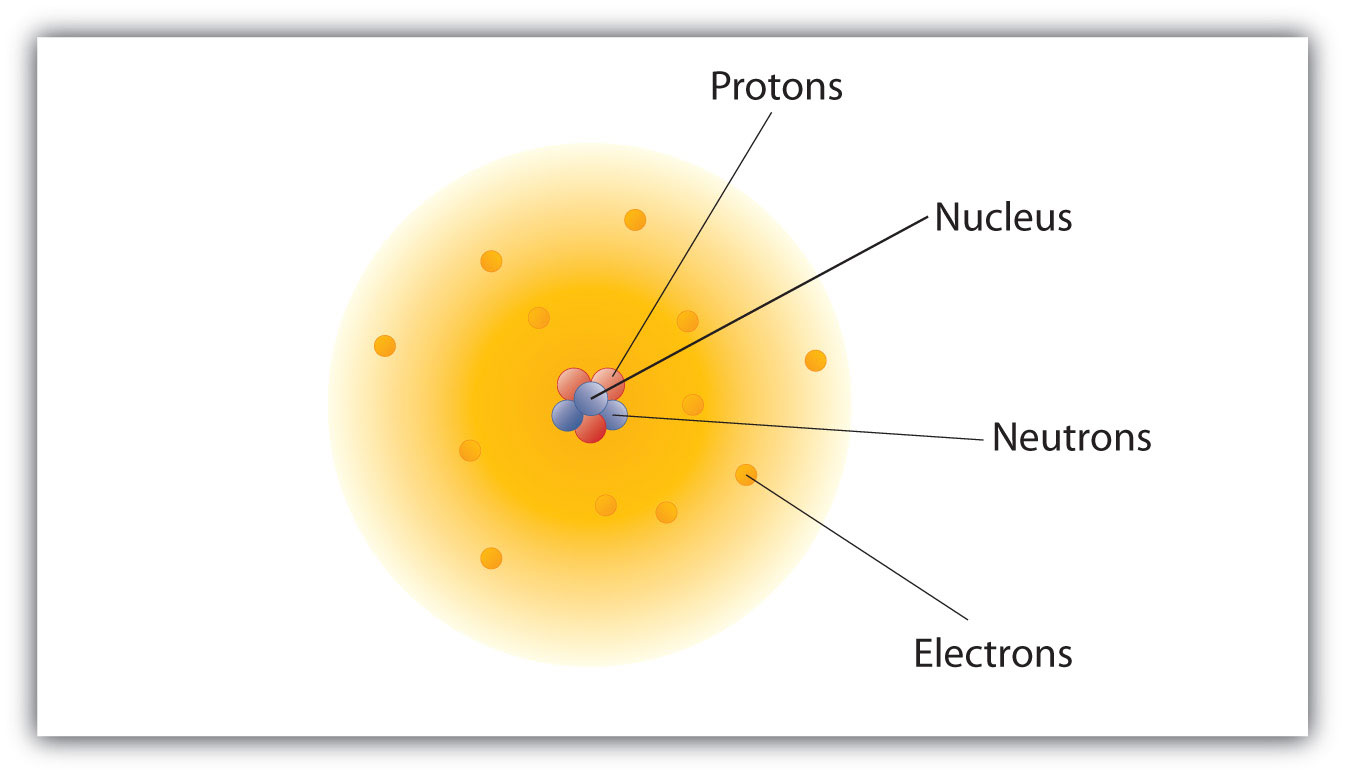
. The modern atomic model represents atoms containing a nucleus of protons and neutrons and a vague gradient or cloud surrounding it containing the electrons. This is the modern atom model. The modern model of the atom describes the positions of electrons in an atom in terms of probabilities.
This model is patterned on the solar system and is known as the planetary model. The modern model of the atom reveals an atom that is mostly empty space with a tiny dense positively-charged nucleus. His new model described the atom as a tiny dense positively charged core called a nucleus surrounded by lighter negatively charged electrons.
An electron can potentially be found at any distance from the nucleus but depending on its energy level and angular momentum exists more frequently in certain regions around the nucleus than others. Which of these statements describe the two models correctly. Most of the space.
Check all of the boxes that apply. The nucleus contains nearly all the mass of an atom. His model was called nuclear model because the center of the atom is a nucleus its very small dense and positively charged also it contains protons and some electrons.
The atom is made of protons neutrons and electrons. The key and not incorrect points of this model are. He proposed that electrons are arranged in concentric circular orbits around the nucleus.
In 1913 Neil Bohr a student of Rutherfords developed a new atom model. Nuclear reactions can alter. The modern model of the atom comprises a dense nucleus at the center of the atom which is made of protons and neutrons.
How is the electron cloud related to the modern model of an atom. Quantum Mechanical Model of Atom. The three parts of the atom are protons positively charged neutrons neutral charge and electrons negatively.
The modern model of the atom is based on quantum mechanics. In the center small nucleus is present made of protons and neutrons. The nucleus is surrounded by a region called the electron cloud.
This is sometimes referred to as the. Ii Atom is made upon mainly of electrons protons and neutrons. The modern atomic theory is described as follows.
Compare the modern electron cloud model of the atom with daltons atomic model. This model is often depicted in artwork showing a central atomic nucleus and oval lines representing the orbits of the electrons. Model of the Atom An atom is a building block of matter that cannot be broken apart using any chemical means.
Daltons model was replaced only because of its age. The modern atomic model is composed of a nucleus in which the protons and neutrons are located and is also surrounded by an electron cloud in which the energy levels that the electrons are on is. The modern model of an atom involve the description of electrons as forming electron clouds and.
Generally speaking the Bohr model encapsulates the modern understanding of the atom. This model which is the basis of the modern understanding of the atom is known as the quantum mechanical or wave mechanical model. Remaining electrons move around the nucleus like are planets around the sun therefore and atom is mostly empty space.
Almost all of the volume of an atom consists of empty space in which electrons the fundamental carriers of. Daltons model represented atoms as indivisible but the modern theory recognizes that they contain. The electrons inhibit their designated energy levels within the electron cloud.
Quantum mechanics is based on Schrödingers wave equation and its solution. But we know that electrons dont really behave like planets orbiting a central star. This model was established following the discovery of quantum mechanical properties such as wave-particle duality and uncertainty.
The modern atomic model involves a dense atomic nucleus containing a fixed number of protons and neutrons surrounded by a probabilistic cloud of electrons. The Modern View of Atomic Structure Almost all of the mass of an atom is contained within a tiny and therefore extremely dense nucleus which carries a. Modern Atomic Theory Models.
Therefore atom no longer remains indivisible. The probability of finding an electron at a point within an atom is proportional to the ψ 2 at that point where ψ represents the wave-function of that electron. The following four principles can summarize the Bohr model.
This model has some good ideas in it but overall it has some problems. We can only describe such particles by saying where they will. Another way of thinking about this model was that the atom was seen to be like a mini solar system where the electrons orbit the nucleus like planets orbiting around the sun.
The Bohr Model contains some errors but it is important because it describes most of the accepted features of atomic theory without all of the high-level math of. However atom is still the smallest. Protons and neutrons form.
I All forms of matter are made upon of atoms which cannot be created or destroyed. The fact that there are only certain allowable states or energies that an electron in an atom can have is similar to a standing wave. This pattern is referred to as its atomic orbital.
Because the Bohr Model is a modification of the earlier Rutherford Model some people call Bohrs Model the Rutherford-Bohr Model. Electrons are in constant motion around the nucleus protons and neutrons jiggle within the nucleus and quarks jiggle within the protons and neutrons. The modified Daltons atomic theory is commonly known as the modern atomic theory.
This picture is quite distorted.

The Development Of The Atomic Model Wired

Timeline Of The Atomic Theory Text Images Music Video Glogster Edu Interactive Multimedia Poste Atomic Theory Interactive Multimedia Chemistry Projects
Comments
Post a Comment